HydroSurf - Hydrophobic Air-water Interface
Prof. Dr. Thomas D. Kühne, Universität Paderborn
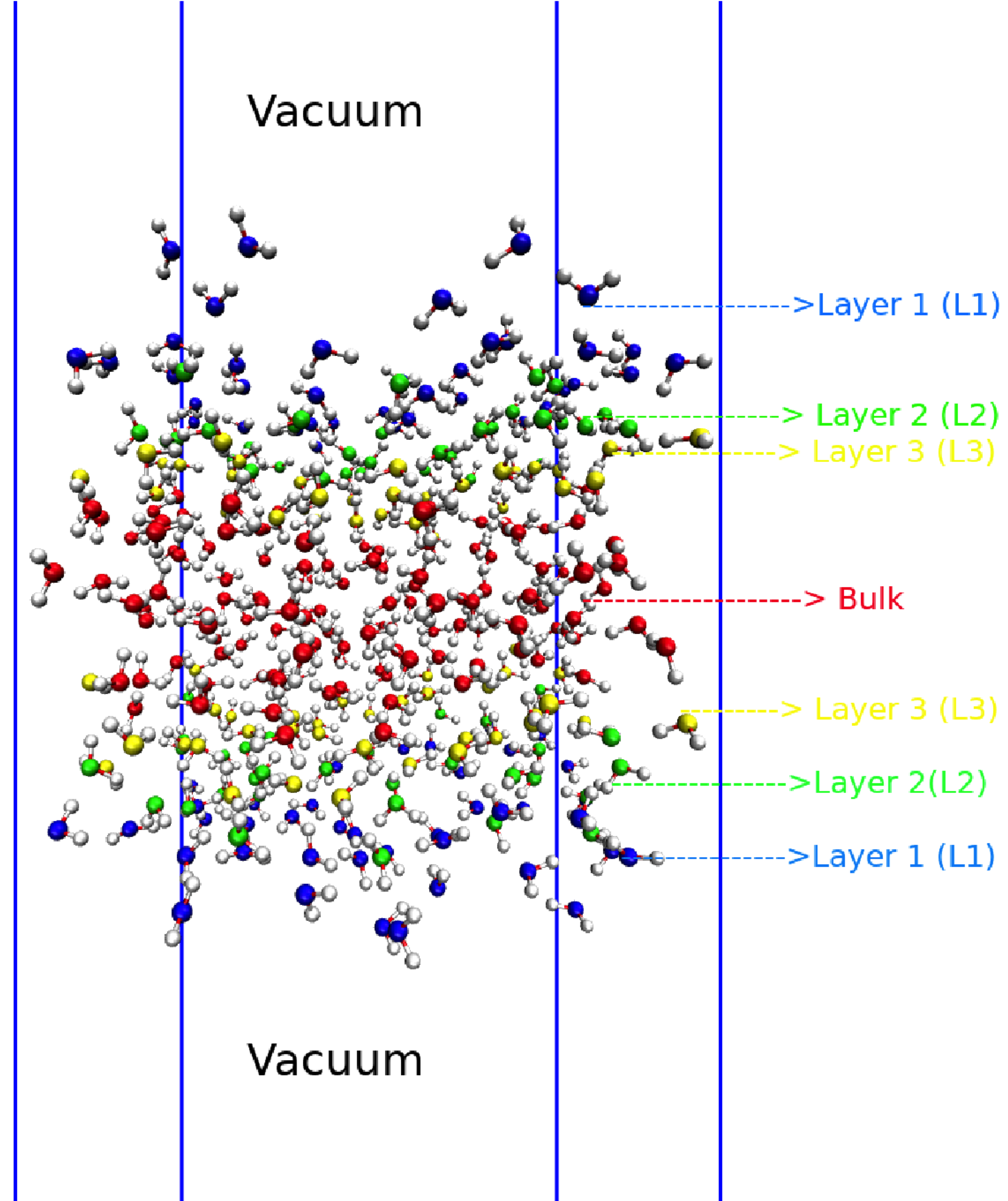
The project is based on the results of recent theoretical and experimental investigations regarding the acidic character of the hydrophobic air-water interface, hinting that proton hopping processes at the air-water interface occur preferentially via water-wires oriented parallel to the surface plane which lead to an increase of the residence time of the proton at the interface, thereby resulting in an interface which is more acidic than that of bulk water.
These water-wires have been shown to be the direct consequence of the specific HB-structure which is formed at the air-water interface in order to maximize water-water H-Bonds, consisting in an extended & continuous 2D-H-Bond-Network, with more than 90% of interfacial water being interconnected through H-Bonds all oriented parallel to the surface. Since the 2D-H-Bond-Network is formed at the hydrophobic air-water interface in order to maximize water-water H-Bonds, similar highly interconnected and preferentially oriented networks are expected to form at other aqueous interfaces, where water is in contact with "sufficiently hydrophobic" surfaces. At these interfaces, if the formation of the 2D in-plane water-wires is verified, then it will lead to the pivotal conclusion that hydrophobic interfaces are systematically acidic regions, with essential consequences for the chemical reactivity at these interfaces.
The validation of this hypothesis is the primary objective of the proposed theoretical work. Once this is achieved through DFT-MD simulations, including quantum nuclei DFT-MD simulations for a complete structural characterization and rationalization of the impact of nuclei quantum effects on the proton hopping mechanism and on the water-wires at hydrophobic aqueous interfaces, then the second objective is the identification of SFG/IR/Raman spectroscopic markers for interfacial protons at these interfaces, which can be in turn be experimentally probed, thereby providing definitive markers for surface acidities still missing in the literature.